How the Elements Affect the Human Body
The purpose of this website is to explain how the elements found on the periodic table of elements affect the human body. Many elements help function and make up the human body while some elements are harmful to the body.
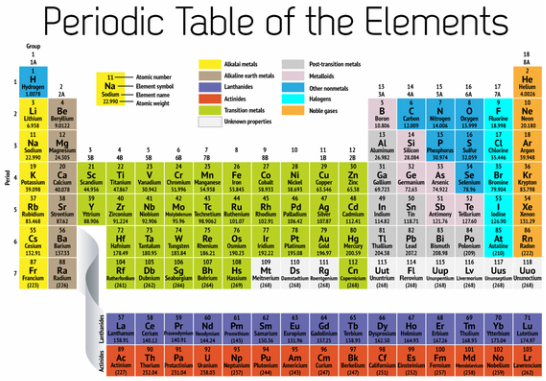
The human body consists relatively of 60 chemical elements. Not all the elements functions in the body are known to scientists. Approximately 96% of the human body is composed of four main elements; oxygen, carbon, nitrogen, and hydrogen. The remaining 4% of the human body consists of other elements found on the periodic table of elements. Every element plays a different role in the human body and helps the body function properly. Although many elements are vital for life, many elements are harmful and in some cases lead to death.
The alkali metals are the elements that make up Group 1 of the periodic table: lithium, sodium, potassium, rubidium, cesium, and francium. Sodium, and Potassium all play major roles in maintaining proper function in the body. Lithium is used to help treat with mental disorders.
The alkaline earth metals are found in Group 2 on the periodic table of elements. Group 2 is comprised of beryllium, magnesium, calcium, strontium, barium, and radium. Beryllium can affect the lungs and skin of a human when they come in contact with it. Magnesium and calcium help keep the body's functions work properly.
The transition elements make up Groups 3 through 12 of the periodic table. These elements, all of which are metals, include iron, cobalt, copper, zinc, chromium, manganese, molybdenum, gold, silver, mercury, nickel, platinum, titanium, vanadium, zirconium, palladium, and tungsten. There are more elements that are considered to be transition metals. Trace elements are elements found in small amounts within the human body and are vital for maintaining health. Also referred to as micro-minerals, trace elements are part of enzymes, hormones and cells in the body. A person can experience nutritional deficiency if there is an insufficient intake of trace minerals.
Nonmetals are the elements that occupy Groups 14-16 on the periodic table. Non-metals are unable to conduct electricity or heat very well and are very brittle. non-metals can exist in two of the three states of matter at room temperature: gases and solids.
The halogen families composed of five chemical elements that make up Group 17 on the periodic table. This family consists of fluorine, chlorine, bromine, iodine, and astatine. The term halogen means "salt-former." Halogens are nonmetallic, have seven valence electrons in their outer shell, and exist in all three states of matter at room temperature: solid- iodine/ astatine, liquid- bromine, gas- fluorine/ chlorine.
The noble gases are the six elements that encompass Group 18 on the periodic table. The noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Tthis family of elements was once known as the rare gases. This lack of reactivity has led to a second name by which they are sometimes known—the inert gases.